 |
| Heat Conduction Page in my Physics-World Software |
CONDUCTION AND INSULATION:
If you coat a knife blade with butter and hold the knife tip near a flame the butter will quickly melt .It melts at the tip first
and then begins to melt further and further from the end .This is because the heat of the flame passes down the length of the blade .The passage of heat through a solid substance , like the knife is called conduction.
The momentum of atoms and molecules within the solid causes conduction.When the molecules near the flame are heated they vibrate(move back and forth) more rapidly .As they vibrate faster , their energy increases.They jostle the molecules next to them and these too move more rapidly .Eventually all the molecules in the solid are vibrating faster and so have more energy .This energy is heat. As it passes down the blade ,the temperature of each part of the knife increases.
THERMAL EXPANSION:
Heating a substance gives its particles more energy so that they move faster and farther. The particles take up more room and increase the volume of the substance .This is known as thermal expansion. Example:
 |
| This cold metal ball passes easily through the hoop |
1) This cold metal ball passes easily through the hoop. The ball is a solid object.Compound of millions of tightly packed vibrating particles.
2) A hot flame heats the ball, giving its particles more heat energy.
EQUATION OF HEAT CONDUCTION:
Q = K * A *(O2 - O1/d)* t
where Q = amount of heat conduction, K= constant, A= area of a material where heat conduct, which is proportional to Q,
O2 - O1 = temperature difference between two plane of the conducting material, which is proportional to Q,
d = density of material, which is inversely proportional to Q,
t = time of heat conduction, which is proportional to Q.
GOOD AND BAD CONDUCTORS:
 |
| Silver is a good Conductor |
Some solids are better conductors of heat than others.Metals such as Copper , Aluminium, and silver are good conductors.Non metals ,including glass wood rubber and plastic do not conduct well .These bad conductors are called insulators.
 |
| Copper Also a Good Conductor |
Materials, such as copper, that are good conductors of heat are usually also good conductors of electricity .Good heat insulators are often good electrical insulators too.
When the knife was held in the flame , the handle did not get hot because most knife handles are made of insulating substances .So are saucepan handles., but saucepans are metal or enamel so that heat can easily pass from cooker into the pan.
 |
| Wood is a Bad Conductor |
Most liquids are bad conductors. Milk can be kept cool in the summer by putting the bottle into a bucket full of cold water.Gases such as air are also very bad heat conductors.Woolen clothings stops heat being lost from the body because there is air trapped between the strands of wool .The layer of air between two jumpers, or the pockets of air in the holes of the strings vest,will make the conductivity even less. This insulation keeps out cold air and prevents heat being lost from the wearer`s body.
Good conductors are used when heat has to flow easily.Insulators are used to make the flow of heat as small as possible. A car radiator is made of metal.The water inside it cools the engine, and therefore gets hot .The metal conducts away this heat. A rubber hot-water bottle filled with hot water will keep warm for a long time.The small amount of heat which passes through the bottle heats the bed.The bed stays warm because hot air is trapped between the blankets. Another example of insulation is the lagging on pipes, hot water tanks,and boilers .It reduces the loss of heat from a hot water system. It also prevents the water in pipes from freezing during a cold water.
 |
| VACUUM FLASK |
VACUUM FLASK:
A vacuum does not conduct at all , this is because there are no molecules there to carry the heat . A vacuum flask is used for keeping thing hot or cold . A glass bottle inside the flask has double walls . All the air is removed from between the walls to leave a vacuum. The walls are silvered to stop heat loss by radiation.
 |
| Internal structure of VACUUM FLASK |
CONVECTION:
 |
| Heat Convection Page in my Physics-World Software |
If watch a saucepan of water being heated you will see ripples of water breaking the surface .These are caused by hot water rising from the bottom .Its place is taken by colder water from the top. This is called convection .It is passage of heat by movement of the material itself.In conduction movement of heat takes place without any movement of material.
Convection occurs in gases as well as liquids. Carefully heat a metal rod(not so hot yhat it burns your fingers) and hold it in the air.Put your other hand a little above it and you can feel the warm air rising from the hot rod. If you put your hand below it, you will not feel any warmth.
Convection takes place because gases and liquids get bigger or expand when they are heated.This expansion makes them less dense than the surrounding material. So they rise and their place is taken by the dense colder fluid from above.These movements of material are called convection currents.The current can be quite strong .Place a small paper windmill above an electric light and watch it spin around.This is how a flickering flame effect is made in some electric fires.
Because the way convection works , a glass of water cannot be cooled by standing it on a block of ice.The water at the bottom becomes colder and colder .It also becomes more and more dense and therefore stays at the bottom .The warmer water at the top has no chance of cooling because it always stays at the top .
CONVECTION CURRENTS:
In the early days of coal mining.The mines were ventilated by convection currents .Two shafts were sunk and a fire lit at the bottom of one of them.As the heated air rose out of this shaft fresh air was drawn into the other one , thus ventilating the mine.
Convection heaters work on the principle that hot air rises. Cold air from the near the floor is taken in and heated.It rises by convection to come out of the top of the heater.A stream of warm air constantly emerges from the heater.Fan heaters and hairdriers transfer heat by convection.Air is heated by an electrical element and blown out by a fan. Heat is being transferred by the movement of air so convection is taking place .When a fan used , it is called forced convection.
Convection currents take place in the atmosphere.The land is heated to a higher temperature than than the sea during the day.Air over the land becomes warmer ,rises, and its place is taken by cooler air from the sea.So there is usually breeze towards the land during the day.At night the land cools down more quickly than the sea so the sea is warmer than the land. A convection current takes place the other way.The air over the sea is heated ,rises and cool air from the land takes its place.
 |
| MINE VENTILATION Picture |
MINE VENTILATION:
Ventilation in coal mine .The fire heats the air which expands , become less dense , and rises out of the shaft .Fresh air comes in the other shaft to ventilate the mine.
 |
| Mine Ventilation Process Detail |
ELECTRIC HEATER:
 |
| ELECTRIC HEATER in Modern house |
There are two types of electric convection heater for rooms. In the natural convection heater the cold air enters the bottom, It is heated by passing over an element , and the warm air rises out of the top .The natural convection current keeps the air circulating .In the fan heater which can be much smaller , a fan sucks it cold air from the back and blows it over the heating elements.
RADIATION:
 |
| Heat Radiation Page in my Physics-World Software |
If you stand in the sunshine on a fine day you will feel warm. But if the sun is suddenly hidden by cloud you will no longer feel its warmth.This means that the sun can warm you without heating the air.Heat is travelling without using the material between you and the sun . Also there is no air beyond the earth`s atmosphere.So the sun`s heat must be able to pass through a vacuum (a space which has no particles in it). This way of transferring heat is called radiation.It is the way that energy travels across empty space .
This is also how heat is passed from the bar of an electric fire .If you stand in front of an electric fire you will feel warm .If you move to one side ,you will feel cooler. Once again the heat is being passed without using the air.Heat radiates from the fire in straight lines, just like light from a torch you can block the light .In exactly the same way someone standing between you and an electric fire blocks the heat and stops it from reaching you.
 |
| Heat Radiation by different colored Objects |
RADIANT HEAT AND LIGHT:
Radiant heat and light are very similar.They are both electromagnetic waves. Radiation is not only emitted by extremely hot things,such as the sun and electric fires.It can also be emitted by cooler objects .If too much heat is allowed to radiate from a teapot then the tea gets cold quickly .Putting a tea cosy over it reduces the radiation loss .
Some surfaces are better radiators of heat than others. Try this experiment with an old baked bean tin.Scrape off the label and paint one half back .Leave the other side shiny .Fill the can with hot water and hold the back of your hand first near the black side and then near the shiny side.You will find that your hand is warmer when held opposite the black side.This is because a black surface is a good radiator of heat . A polished surface is a bad radiator .Repeat the experiment with another can, whose sides are painted white and brown.Generally , a darker surface radiates more heat than a lighter one.
From these experiments you will understand why a silver teapot with highly polished sides will not lose much heat by radiation. Whereas a brown or black teapot will lose much more.
As well as radiating heat, surfaces can also reflect and absorb heat.When it falls on a surface , heat is partly reflected and partly absorbed , just like light .Good radiating surfaces are also good absorbers of radiation .Bad radiating surfaces tend to reflect heat instead of absorbing it.
People who live in hot countries do not want their clothes to absorb heat .They therefore wear white clothes and hats which reflect the heat.Space craft have highly polished surfaces to reflect away the radiated heat from the sun.
The sun radiates both heat and light to the earth.You can see the light rays which are radiated but you cannot see the heat rays.You can only feel them as they warm you up.
 |
| Firemen wear suits with highly polished surfaces so that the heat from the fire reflected and not absorbed |
 |
| n cold Arctic region Eskimos were live in the house made of polished ice called Igloo. |
In cold Arctic region Eskimos were live in the house made of polished ice called Igloo. Polished ice radiate very little heat ,which keep warm inside the Igloo.
 |
| Refrigerator Page in my Physics-World Software |
LOW TEMPERATURES:
It is fairly easy to produce high temperatures.Making things colder is more difficult.One way of producing low temperatures is by evaporating the liquids. You can demonstrate this by wetting your hand and waving it around.Your hand will feel colder.This is because the water is turning into vapour.
when water boils it needs heat to change into vapour.In the same way the water on your hand needs heat to evaporates the more heat is taken away. Try the same experiment with methylated spirits or after-shave lotion which both evaporate more quickly than water.
REFRIGERANTS:
Inside a refrigerator there are tubes containing either a liquid with a very low boiling point or a gas that can easily be changed into liquid under pressure .It is called refrigerant.Some examples are ammonia.ethyl chloride, and freon.The refrigerant is first compressed to a high pressure in one part of the refrigerator.
 |
| Temperature of Cooled Ice |
At this high pressure the refrigerant`s molecules are pushed close together so it is a liquid.The compressed liquid is passed through a valve into a region of low pressure .At this low pressure the liquid changes back into a vapour.This fast evaporation takes heat from the inside of the refrigerator, just as the water evaporating took heat from your hand.The refrigerant is compressed to turn it back to a liquid and used again.The heat passes out through the back of the refrigerator.
 |
| Domestic Refrigerators |
Many domestic refrigerators use evaporation to cool food and make ice .
A slightly different method is used to produce very low temperatures.It depends on the fact that if a gas expands quickly it cools when you pump up a bicycle tyre you are compressing the air . You have probably noticed that the pump gets hot when you do this .The opposite happen when a gas at high pressure is suddenly changed to low pressure. The gas gets slightly colder.
This slight fall in temperature is called the Joule-Kelvin effect after its discoverers James Prescott Joule and lord Kelvin.Using this effect , it is possible to cool gas so much that it becomes liquid .Oxygen changes from a gas to a liquid at -183 0 C.Nitrogen gas changes to liquid at - 195.8 0 C.
These are very low temperatures,but even lower temperatures can be produced.
 |
| In Very Cold Mountain Country Water lake Become frozen in Winter. |
ABSOLUTE ZERO:
The temperature of anything depends on how fast the atoms or molecules are moving about .If they are moving very quickly the temperature is high . If they are moving slowly the temperature is low .At the lowest temperature the atoms and molecules are not moving at all .It is called absolute zero and is - 273.15 0 C .Scientist have produced temperatures that are only a millionth of a degree above absolute zero but they have never reached it.
REFRIGERATOR:
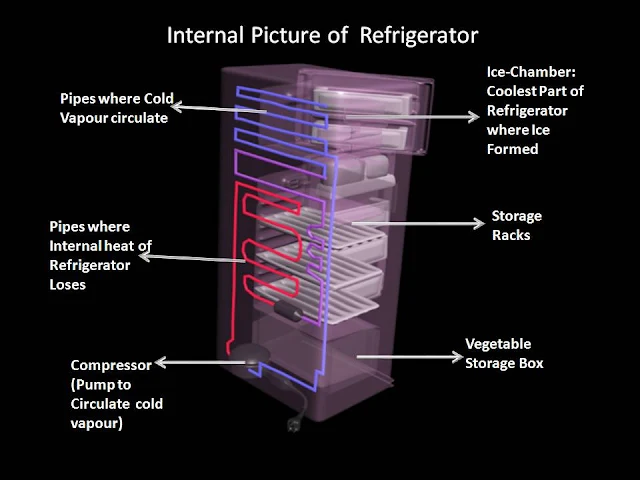 |
| Internal Structure of Refrigerator |
In an ordinary refrigerator the coldest part is the freezer. This is below 0 degree centigrade and can be used for making ice and keeping frozen food. The rest of the refrigerator is just above 0 degree centigrade and is used for keeping food fresh. Inside the refrigerator the Refrigerant is turned to a liquid by compressing it and heat is given out, after it passes through the expansion valve , this liquid turn to a gas and cools , taking heat from the refrigerator and its contains.

























 Online Movies
Online Movies
Amazing blog! Are you looking for the best printed Personalised Flask? so you should visit our website & here you can see the all types of personalised flask, Insulated Steel Travel Bottle, etc.
ReplyDeleteYes, indeed. There is no reason to doubt the validity of this view. Many technologies have already applied this idea at least once. So, for the next promotional event if the company is looking for smart but impressive gifts, then promtional pen
ReplyDeletecan be a reliable proposal. This is also economical, the most important part of your budget will be saved.